Over the past six weeks, the team of three—Stephen Holler, Ph.D., associate professor of physics; Alan Vara, a rising senior from the Bronx; and Bryant Ramos, a rising senior from Brooklyn—have been collaborating on climate change research through the Fordham Science and Technology Entry Program (STEP), designed to help underrepresented students in junior and high school prepare for STEM-related careers. Inside Holler’s lab, they controlled a carbon dioxide sensor, calculated CO2 concentrations, and dissected their data. At the annual Calder Summer Undergraduate Research Symposium on Aug. 8, they presented their research results alongside six other STEP students, who developed projects related to biology, psychology, and chemistry.
But what seems to have had the largest impact on them, said Vara and Ramos, was learning about the increasingly devastating effects of climate change. Recent historically high carbon dioxide measurements at Hawaii’s Mauna Loa Observatory have made them consider the impact of CO2 in their own homes in New York City.
“You can see that it’s not just there—it’s our neighborhoods as well,” Ramos said.
“It’s a lot more personal. You realize that it’s not [just]in some far off island,” Vara chimed in. “It’s right here, where you live and enjoy your life.”
Dissecting the Science
In mid-July, the two high school students began visiting the Rose Hill campus several times a week.
“Every day they’d come in, and we’d sit down, get the software up and running, start building the box,” Holler said, referring to the box that held their carbon dioxide sensor. “[Then] we’d do some machining and programming.”
The trio’s main tool was a non-dispersive infrared (NDIR) sensor: a special device that converts light rays into numbers. The sensor sucks air into a tube and shines infrared light through the gas. The infrared light comes from a lamp at one end of the tube and shines through to detectors at the other end. A pump sucks in the air from the outside into the tube.
At the other end are two detectors that measure the intensity of the light coming through. What Holler and his two mentees were concentrating on, said the professor, was the way the light interacted with carbon dioxide molecules.
“[CO2] is one carbon and two oxygens. They’re bound to each other. You can think of them as being little masses on springs. They vibrate at certain frequencies. And when your light source corresponds to that frequency that they like to vibrate at, you’ll get light absorption,” Holler said.
In other words, the carbon dioxide absorbs some of that light, thus removing the light from inside the tube. Meanwhile, one detector at the back end measures how much light was absorbed. The other detector measures the amount of remaining light.
“Based on those relative intensities, I can say something about how much CO2 is in the tube,” Holler explained.
The Earth’s rising carbon dioxide levels are primarily the result of fossil fuel burning. This leads to rising sea levels and other phenomena that can negatively affect communities and creatures around the world, as Holler’s high school students have observed this summer. Although the air at the Rose Hill campus is safe, they wanted to take a look at how the carbon dioxide levels fluctuate, particularly during morning and evening rush hours, said Holler.
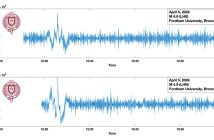
The team ran the NDIR sensor several times throughout the summer: twice in the lab overnight, and a few times in the lab for an hour and a half. Sometimes they threaded a tube from the box through a lab window outside; other times they measured CO2 levels in the lab itself. Then they graphed the CO2 concentrations and analyzed their results.
They noticed that during the morning and evening rush hours, there was a slight increase in carbon dioxide. When the lab’s air-conditioning unit shut down at night, there was an increase in carbon dioxide levels due to the warmer temperatures, as opposed to during the day with cooler temperatures. Their indoor vs. outdoor data were relatively similar.
Overall, the results of their preliminary experiment have prepared them for final data collection tests in the future, Holler said.
“[This shows] that we can have an instrument that monitors CO2 in here,” he said. “We know we have some improvements to make in the way we collect the data.”
But ultimately, Holler wants to create a local network of CO2 sensors that could spark more dialogue about climate change. Instead of having people hear about data from distant places like Hawaii, he wants them to see what’s happening right in their neighborhoods—and realize that this issue does impact them, Holler said.
“What I want to do is really have a tool for communicating—that we can engage non-scientists, we can engage the public on these issues that we’re dealing with right now,” he said.
Bringing Climate Change Closer to Home
Ramos said he knows that climate is real. But it was surreal to learn about the issue up close—to learn how a local sensor could capture carbon dioxide data in his own city.
“We were talking about the environment and stuff at school,” said Ramos, who attends the Manhattan Center for Science and Mathematics in East Harlem. “I can bring this to school and share it [with people].”
Vara, a rising senior at the High School for Mathematics, Science, and Engineering at City College echoed the same sentiment.
“We got the device up and running, but then there was also a why to it. We had to understand why we were building it, what the point of it was, what we were trying to convey in graphing data,” said Vara. “We [also]saw videos about Hurricane Sandy and how it affected Staten Island and how just a couple of centimeters of water, like sea-level elevation, has a disastrous effect for the people on the coast. Getting to see that climate change isn’t as distant as a lot of people would think—it’s not a 100 years into the future; it’s something that’s affecting people right now—that was definitely eye-opening.”